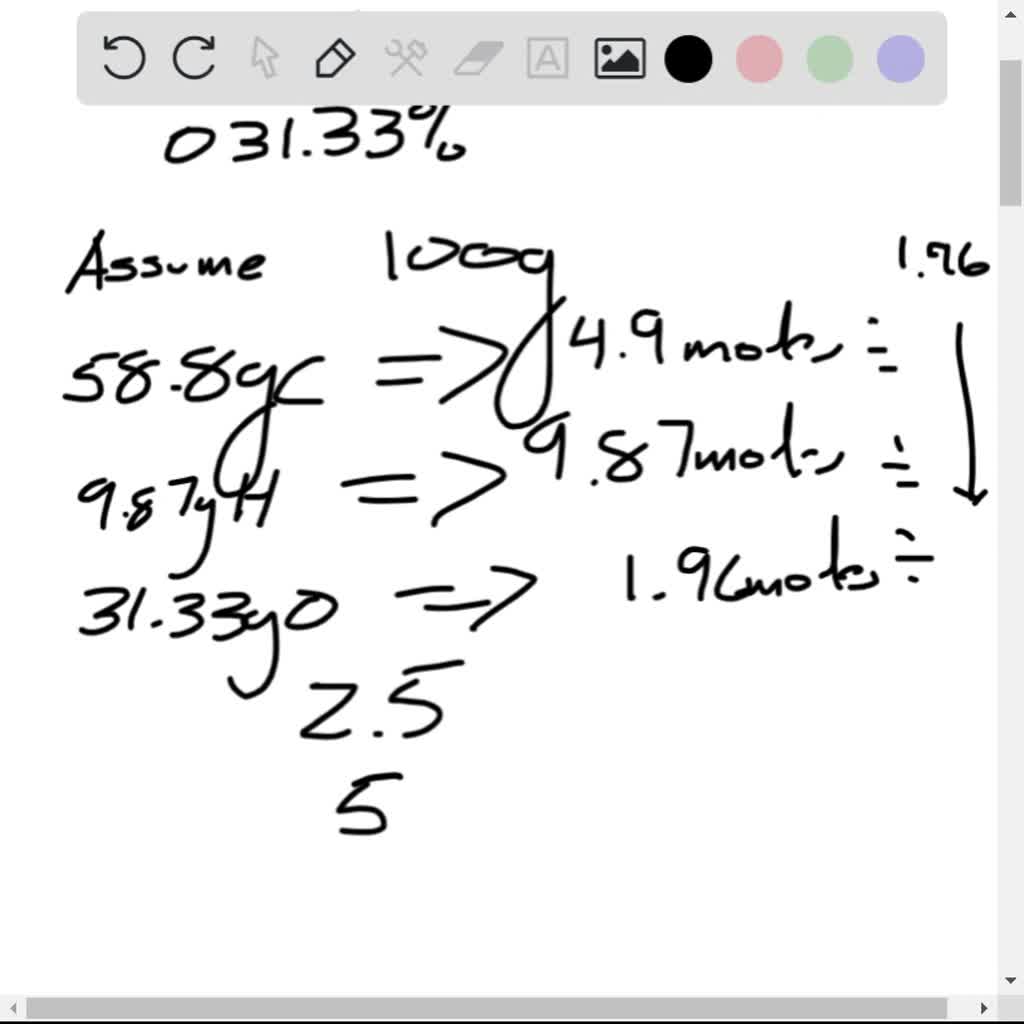
The percent composition tells you how much of each element is present:īecause it is convenient. Start the problem with the assumption that you have 100.0 g of sample.  
to moles.Why a conversion to moles? Because that is what a formula is, aformula relates moles of one element to moles of another element. The problem is really just a conversion from % by wt. The result of an elemental analysis is alwaysreported as a percent mass of each of the elements for which theanalysis was performed.Īn elemental analysis of a white crystalline compound believed tobe C 6H 12O 6, analyzed for C, H and Ogave the following results.Ĭ = 40.00 %, H = 6.71 %, O = 53.28 % by wt. When an elemental analysis is performed we do not get themolecular formula.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
